Nanoscale 'computer' controls function of protein, influences cell behavior
rael-science
Nanoscale 'computer' controls function of protein, influences cell behavior
by Pennsylvania State University
The
creation of nanoscale computers for use in precision health care has
long been a dream of many scientists and health care providers. Now, for
the first time, researchers at Penn State have produced a nanocomputing
agent that can control the function of a particular protein that is
involved in cell movement and cancer metastasis. The research paves the
way for the construction of complex nanoscale computers for the
prevention and treatment of cancer and other diseases.
Nikolay
Dokholyan, G. Thomas Passananti Professor, Penn State College of
Medicine, and his colleagues—including Yashavantha Vishweshwaraiah,
postdoctoral scholar in pharmacology, Penn State—created a
transistor-like 'logic gate,' which is a type of computational operation
in which multiple inputs control an output.
"Our
logic gate is just the beginning of what you could call cellular
computing," he said, "but it is a major milestone because it
demonstrates the ability to embed conditional operations in a protein
and control its function, said Dokholyan. "It will allow us to gain a
deeper understanding of human biology and disease and introduces
possibilities for the development of precision therapeutics."
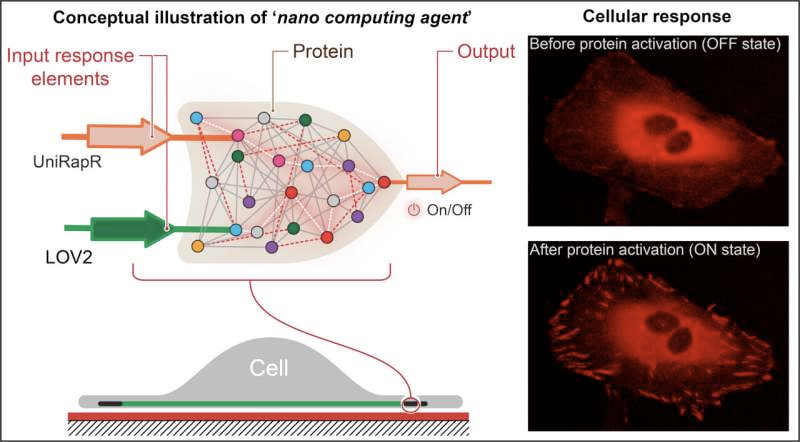
The
team's logic gate comprised two sensor domains designed to respond to
two inputs—light and the drug rapamycin. The team targeted the protein
focal adhesion kinase (FAK) because it is involved in cell adhesion and movement, which are initial steps in the development of metastatic cancer.
"First,
we introduced a rapamycin-sensitive domain, called uniRapr, which the
lab had previously designed and studied, into the gene that encodes
FAK," said Vishweshwaraiah. "Next, we introduced the domain, LOV2, which
is sensitive to light. Once we optimized both domains, we combined them
into one final logic-gate design."
The team inserted the modified gene into HeLa cancer cells and,
using confocal microscopy, observed the cells in vitro. They studied
the effects of each of the inputs separately, as well as the combined
effects of the inputs, on the cells' behavior.
They
discovered that not only could they rapidly activate FAK using light
and rapamycin, but also that this activation resulted in the cells
undergoing internal changes that enhanced their adhesive capabilities,
which ultimately decreased their motility.
Their results published today (Nov. 16) in the journal Nature Communications.
"We
show for the first time that we can build a functioning nanocomputing
agent within living cells that can control cell behavior," said
Vishweshwaraiah. "We also discovered some interesting features of the
FAK protein, such as the changes it triggers in cells when it is
activated."
Dokholyan noted that the team hopes to eventually test these nanocomputing agents in vivo within living organisms.
Other Penn State authors on the paper include Jiaxing Chen, graduate student; Venkat R. Chirasani, postdoctoral fellow; and Erdem D. Tabdanov, assistant professor of pharmacology.
Explore further
